 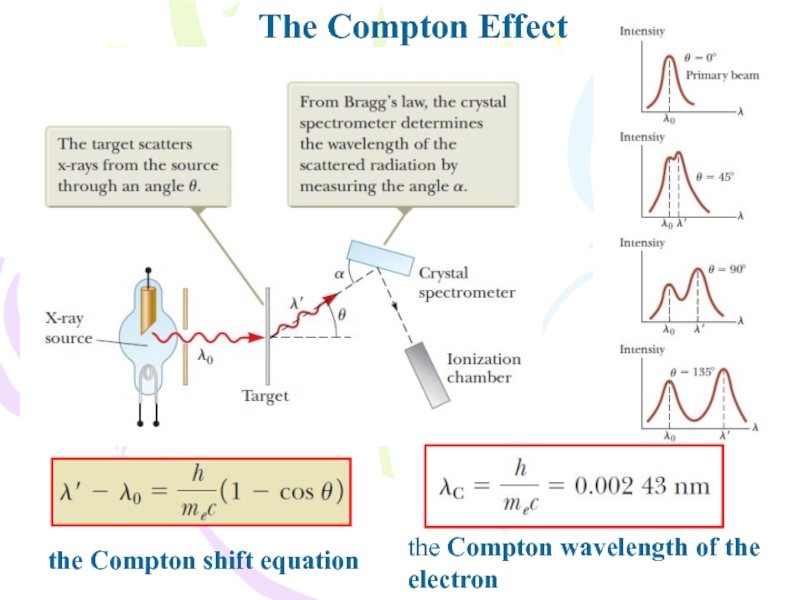
He observed that X-rays scattered in paraffin have a longer wavelength than that of incident rays. In contrast, the Compton effect, was discovered by American physicist Arthur Holly Compton, in 1923. The discovery, upon further investigation, caused a major upset on the prevailing hypothesis that light is a wave and gave rise to the theory that light exists as a particle and wave. This verified that electromagnetic waves of wavelength shorter than those of visible light was irradiating the first gap and causing it to break down. He noticed that the gap between a pair of oppositely charged electrodes broke down, or sparked, quite readily when a second spark gap was fired in the immediate neighborhood. It was by accident that he discover it the phenomenon by accident while conducting some of his monumental studies on the electromagnetic wave. Another photon that is of lesser energy, then moves off at an angle to the direction of motion of the primary photon.ĭiscovery of the photoelectric effect phenomenon was by German physicist Heinrich Hertz in 1887. When a primary photon collides with an electron, some of the energy of the photon is transferred to the electron, which is then ejected from the atom. The Compton effect, on the other hand, is the scattering of photons from gamma or X rays by the electrons orbiting around the atoms. In an electric circuit consisting of a photocell, when electromagnetic radiation, such as ultraviolet rays is incident on a photocell, an electric current flows and the current is called photocurrent. Philadelphia, PA: Wolters Kluwer Health 2012.The photoelectric effect refers to the emission of electrons when electromagnetic radiation is incident on a metal surface. Reprinted with permission from Fosbinder RA, Orth D. As the atomic number of the absorber decreases, the probability of characteristic X-ray emission decreases. The difference in binding energy is released as either characteristic X-rays or Auger electrons. This will, in turn, create another vacancy, which is filled from an even lower binding energy shell, creating an electron cascade from outer to inner shells. An electron from a more outer shell, with a lower binding energy, will fill this vacancy. As a result of a photoelectric interaction, atoms are ionized with a vacancy of an inner shell electron. For example, photons whose energies exceed the K-shell binding energies will most likely result in photoelectric interactions with the K-shell electrons. For a photoelectric effect to occur, the incident photon energy must be equal to or greater than the binding energy of the ejected electron. Illustration of the photoelectric effect.For example, an iodine atom which has a K-edge of 33.2 keV is six times more likely to have a photoelectric interaction with a 33.2 keV X-ray photon than with a 33.1 keV photon ( Figure 2.4). Every element has one or a few sharp “absorption edges” in which the probability of a photoelectric interaction dramatically increases for photons of energies just above the absorption edge relative to energies just below the edge. As the incident photon energy increases to just above the binding energy of the K shell, the absorption of the photon markedly increases. However, there are sharp discontinuities to be observed at certain photon energies. Also, the probability of photoelectric absorption generally decreases with the increase in photon energy. This is why barium ( Z = 56) and iodine ( Z = 53), which have high atomic numbers, are used as contrast agents in X-ray imaging. Therefore, as the atomic number increases, the photoelectric absorption effect becomes more pronounced. The probability of photoelectric absorption, by approximation, is considered proportional to Z 3/E 3, where Z is the atomic number and E is the energy of the incident photon. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
